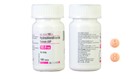
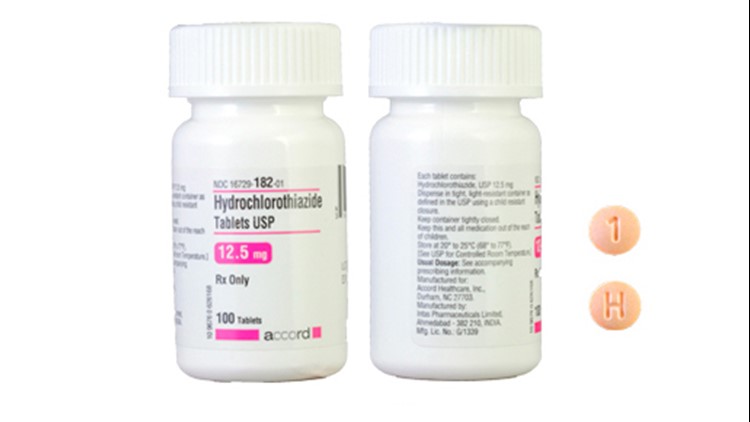
A specific lot of blood pressure medication has been recalled after a bottle from that lot was found to be mislabeled, according to a Food and Drug Administration statement.
The bottle was supposed to contain Hydrochlorothiazide Tablets USP 12.5 mg — a medication used to treat high blood pressure. Instead, they contained Spironolactone Tablets USP 25 mg — used to treat congestive heart failure, cirrhosis of the liver and other ailments.
The FDA says that the effects of mixing up the medications depends on the individual, but can range from "limited" to "life-threatening."
Accord Healthcare Inc. voluntarily recalled a single lot of the drug, PW05264, after investigations revealed it was the only lot that was possibly affected.
Accord’s Hydrochlorothiazide Tablets USP 12.5 mg can be identified by a visual guide. The tablets are supposed to be light orange or peach in color and round. They have a "H" and a "1" on opposite sides.
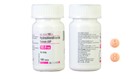
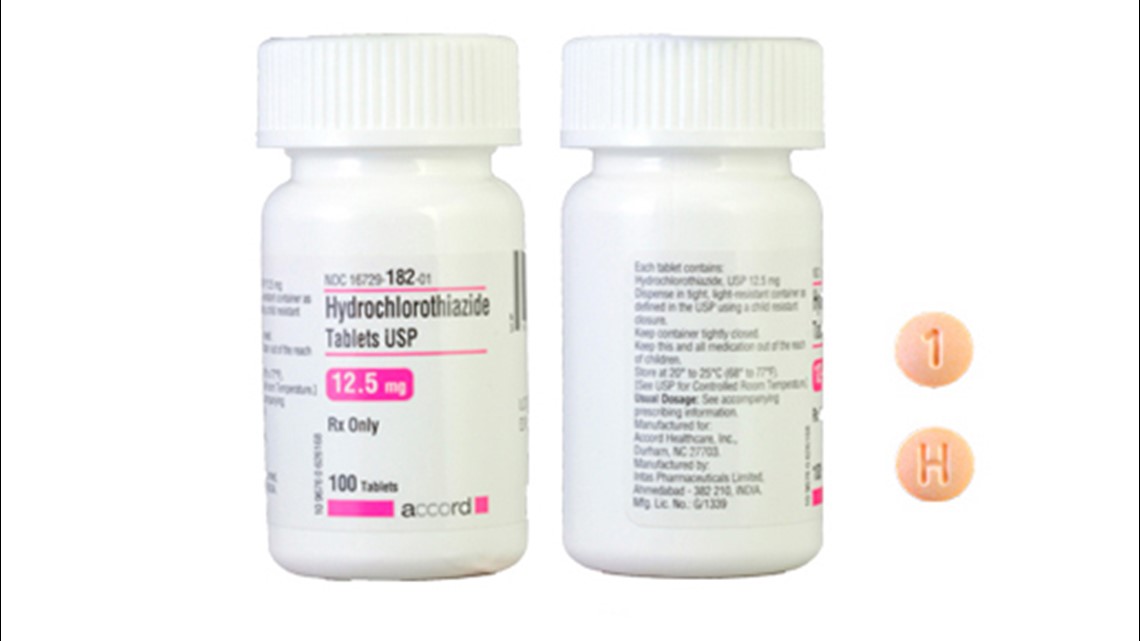
For people in possession of Accord Hydrochlorothiazide tablets that do not match this description, the FDA recommends checking with a pharmacy or healthcare provider.
Accord says there have been no reports of adverse incidents relating to this recall, according to the FDA release.